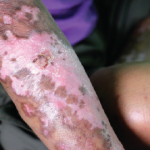
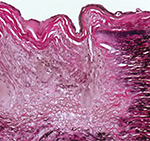
Scleroderma, or systemic sclerosis (SSc), is an autoimmune disease characterized by vasculopathy and fibrosis. Although relatively rare, with a prevalence in North America of approximately 300 per 1 million people, SSc is associated with significant morbidity and high rates of mortality.1 Patients with scleroderma have four times greater mortality than age- and sex-matched controls, with…