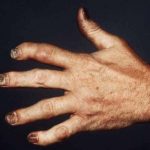
CHICAGO—Bimekizumab is an investigational interleukin (IL) 17A and IL-17 neutralizing agent being studied to modulate inflammation in psoriasis, psoriatic arthritis and ankylosing spondylitis. During the late-breaking abstract session at the 2018 ACR/ARHP Annual Meeting, researchers reported on the results of a 48-week, Phase 2b, randomized, double-blind, placebo-controlled study of bimekizumab in patients with active psoriatic…