Data from a single-center registry shines light on 20 years of trends in first-line, biologic disease-modifying anti-rheumatic drug prescriptions for patients with rheumatoid arthritis.

Subcategories:AnalgesicsBiologics/DMARDs

Data from a single-center registry shines light on 20 years of trends in first-line, biologic disease-modifying anti-rheumatic drug prescriptions for patients with rheumatoid arthritis.
Research has shown voclosporin in combination with MMF and low-dose steroids benefits patients with lupus nephritis, significantly increasing the speed of remission. New data from an ongoing extension study demonstrate a positive risk/benefit profile.
Brandon May |
NEW YORK (Reuters Health)—A significant proportion of children with rheumatic diseases develop new-onset hypogammaglobulinemia and infections following treatment with the monoclonal antibody rituximab, according to new U.S. research. “Increased risk appeared to be mediated, at least in part, by exposure to pulse dose corticosteroids,” write Marc Natter, MD, of Harvard Medical School and Boston Children’s…
Reuters Staff |
(Reuters)—Europe’s drugs regulator said on Monday it was evaluating the use of Roche’s arthritis drug, tocilizumab, in hospitalized adults with severe COVID-19, its latest review of a potential coronavirus treatment. Tocilizumab, sold by Roche as Actemra and RoActemra, has shown promise in clinical trials in treating COVID-19, and was approved by U.S. health regulators in…

ATLANTA—The ACR is actively engaged with the U.S. Food & Drug Administration (FDA) Center for Drug Evaluation and Research (CDER) drug shortage team as they work with the manufacturer to resolve current shortages of tocilizumab (Actemra). Demand for tocilizumab has outpaced supply, with demand increasing after the FDA’s June 24 Emergency Use Authorization (EUA) for…

One of the oldest treatment options in rheumatology, colchicine, may be an effective and inexpensive treatment to prevent complications in non-hospitalized patients with COVID-19, particularly in men, according to the results of large global study.

In July, the FDA approved the use of Octagam 10%, an intravenous immunoglobulin solution, to treat dermatomyositis in adults after an international study demonstrated the treatment’s safety and efficacy.
ATLANTA—The American College of Rheumatology (ACR) today applauded the U.S. Centers for Medicare & Medicaid Services’ (CMS) decision to withdraw a proposed rule to implement the “Most Favored Nation” (MFN) payment model for Part B drugs. Leaders felt the policy would have dramatically disrupted patient access to critical therapies needed to manage rheumatic diseases and…
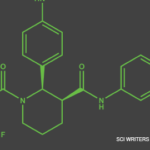
On May 6, the U.S. Food & Drug Administration’s (FDA’s) Arthritis Advisory Committee narrowly voted in support of avacopan, a C5a receptor inhibitor, for the treatment of adult patients with anti-neutrophil cytoplasmic antibody (ANCA) associated vasculitis. Although the panelists were excited about the possibility of a steroid-sparing therapy, some raised questions about whether results from…

Ibrahem Salloum, MD, & Deepan S. Dalal, MD, MPH |
Discovered more than 3,000 years ago, colchicine is one of the oldest drugs still in use today. Like most old remedies, colchicine is a chemical substance found in many plants, most notably in colchicum autumnale, known as wild saffron or autumn crocus. It was mentioned in the oldest Egyptian medical text, Ebers Papyrus (circa 1550…